GMP Tiered Cell Banking of Non-enzymatically Isolated Dermal Progenitor Fibroblasts for Allogenic Regenerative Medicine | SpringerLink

Figure 16 from Mise en place et développement d'une plateforme de culture pour la levure pichia pastoris | Semantic Scholar

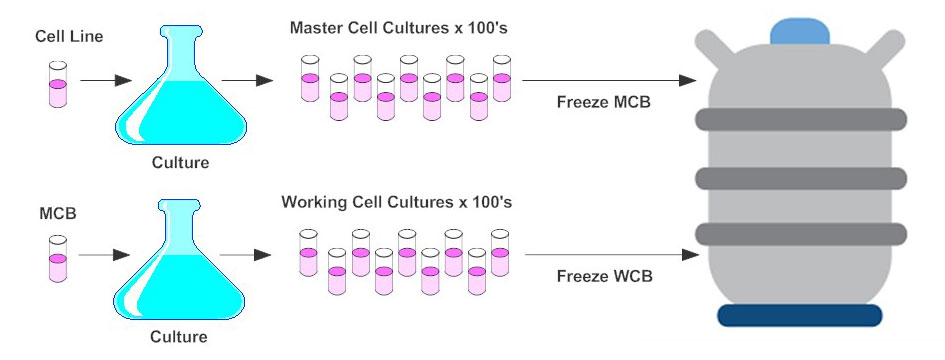
A Roadmap for the Production of a GMP-Compatible Cell Bank of Allogeneic Bone Marrow-Derived Clonal Mesenchymal Stromal Cells for Cell Therapy Applications | Stem Cell Reviews and Reports

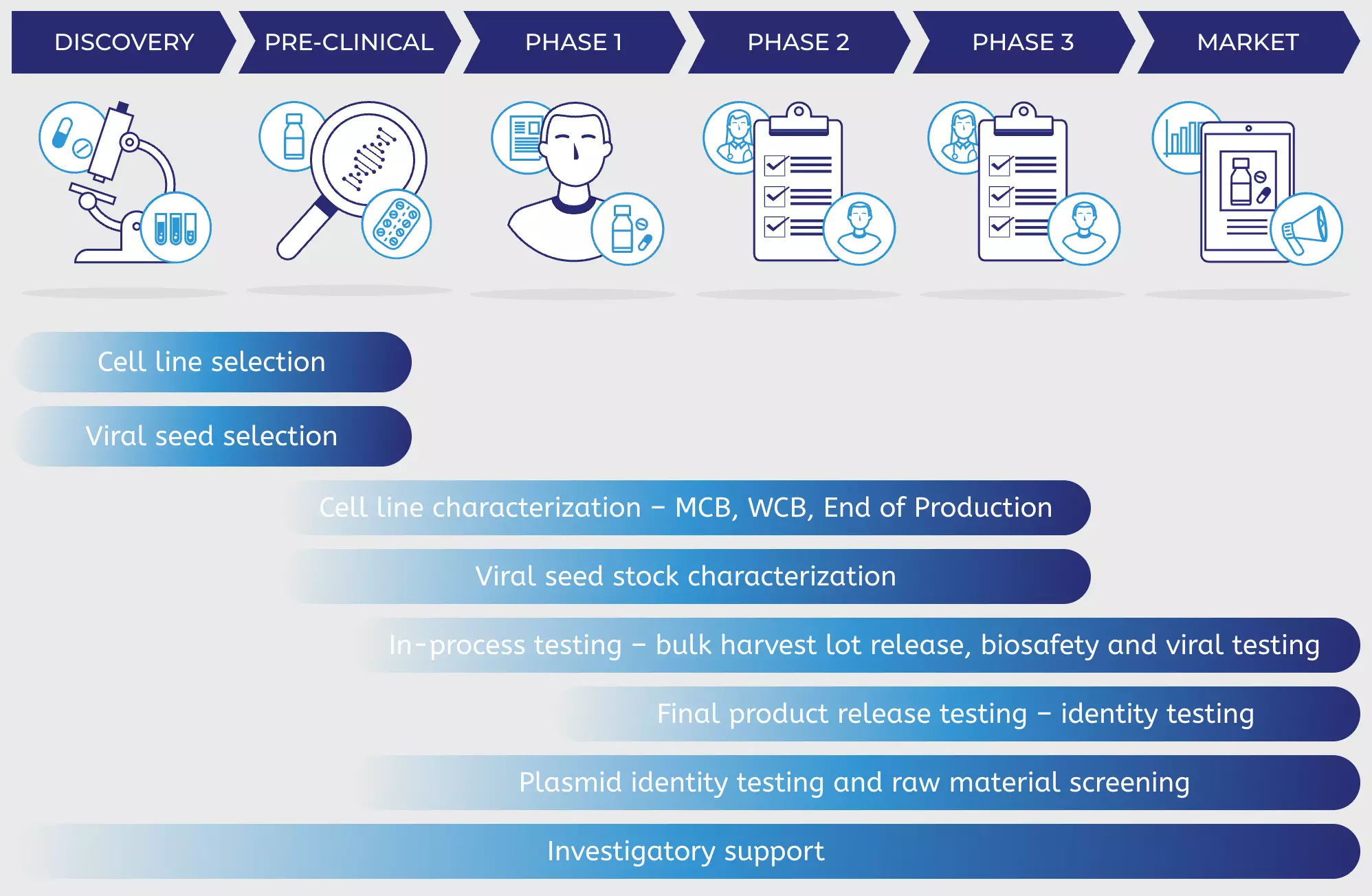
Upstream Bioprocessing - From Pre-clinical to Manufacturing | Biopharmaceutical Manufacturing | MilliporeSigma
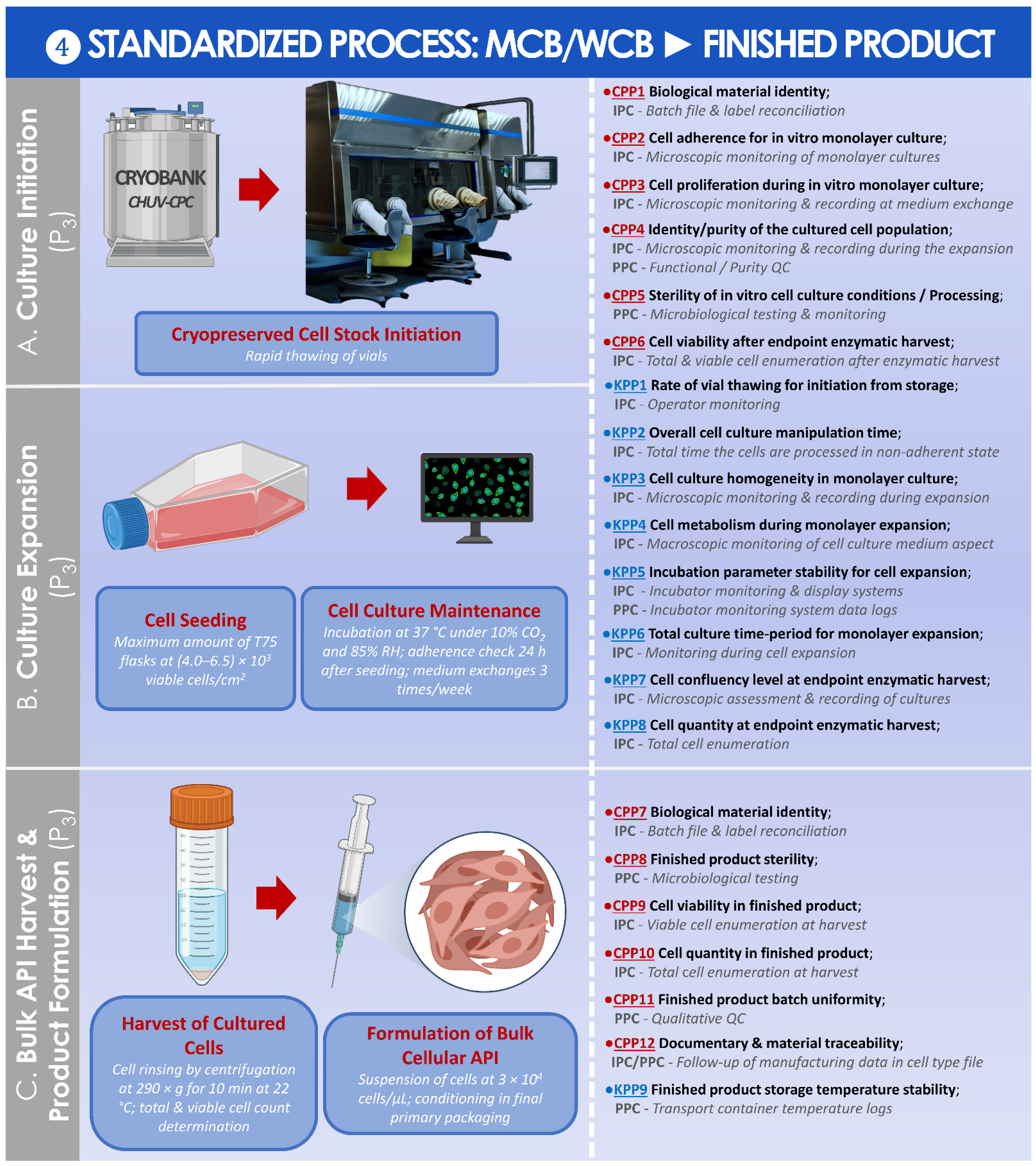
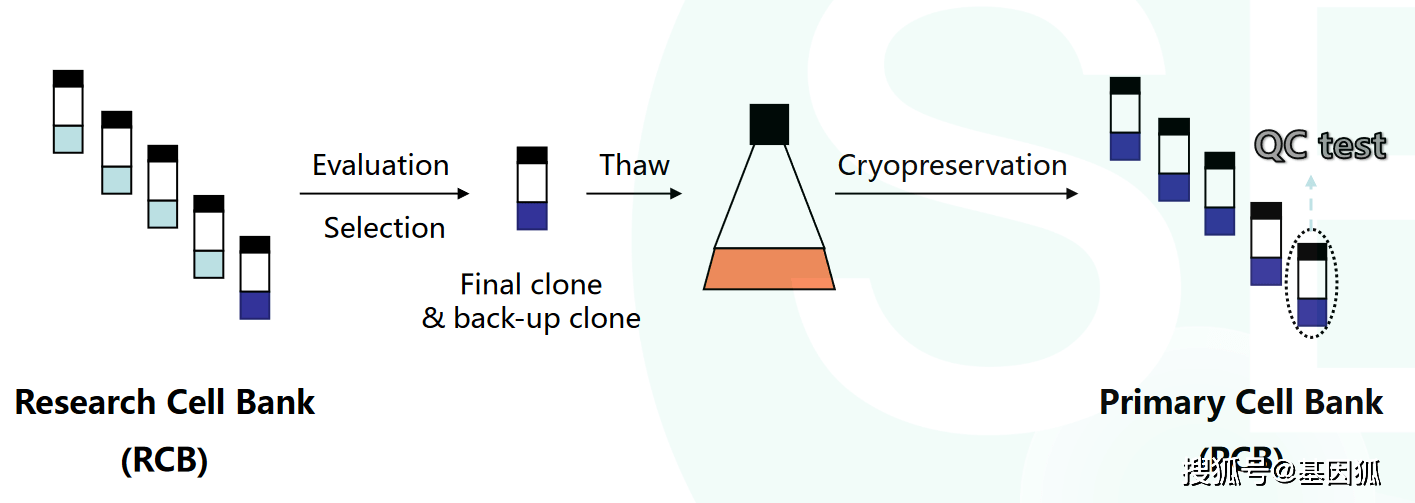
Cells | Free Full-Text | Retrospective Analysis of Autologous Chondrocyte-Based Cytotherapy Production for Clinical Use: GMP Process-Based Manufacturing Optimization in a Swiss University Hospital

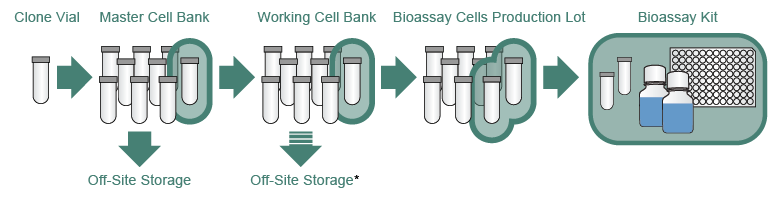
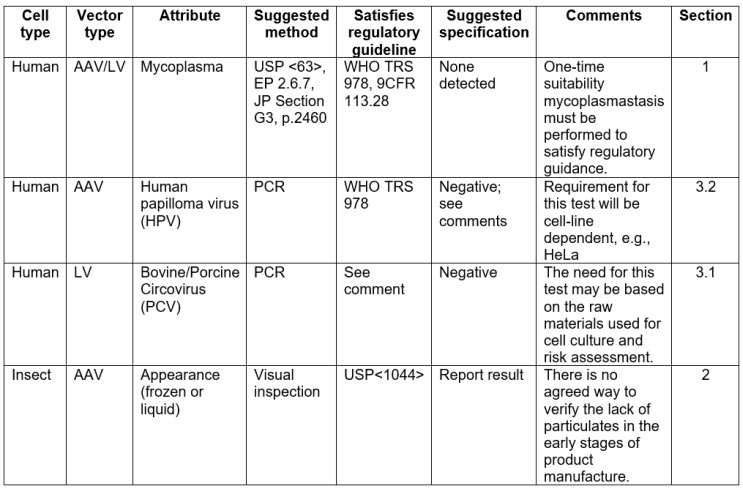
Cell Banking of HEK293T cell line for clinical-grade lentiviral particles manufacturing | Translational Medicine Communications | Full Text
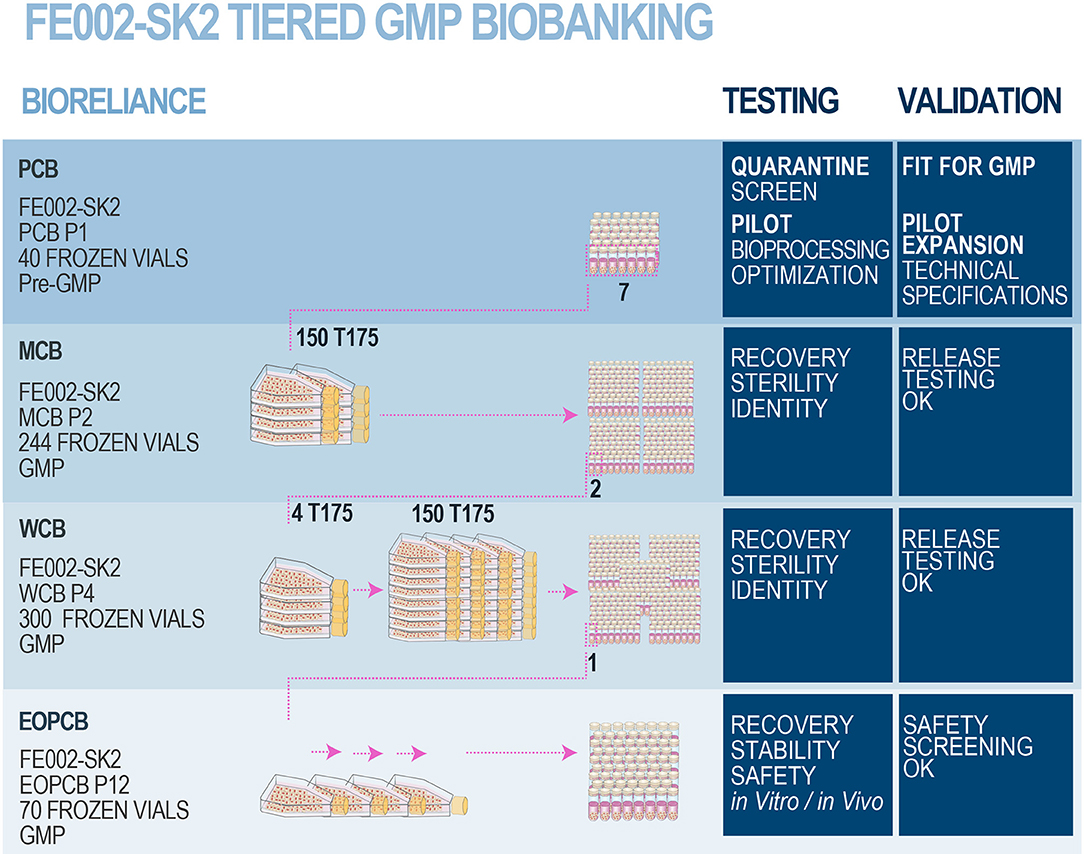
Frontiers | Bringing Safe and Standardized Cell Therapies to Industrialized Processing for Burns and Wounds