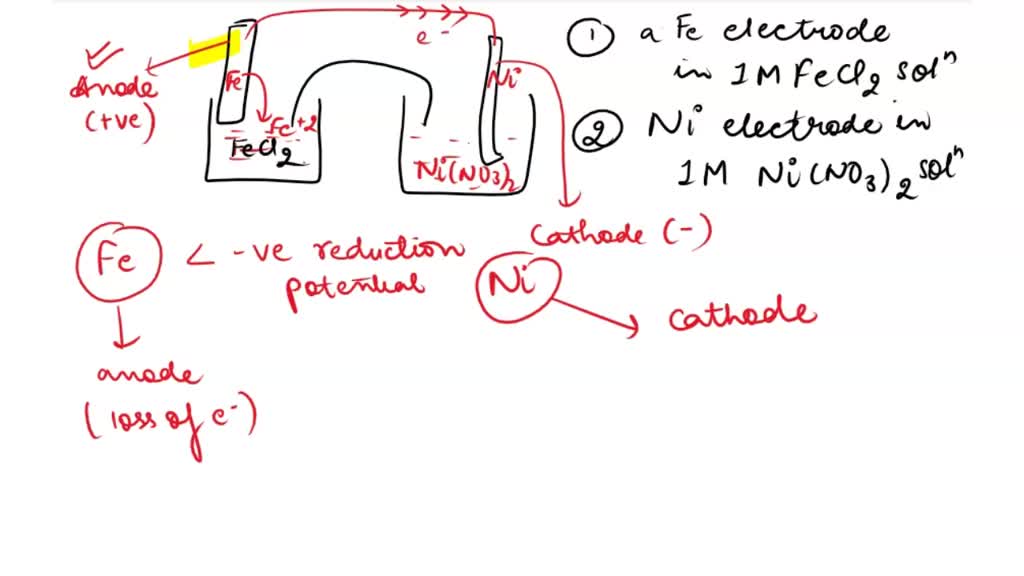
SOLVED: Consider an electrochemical cell constructed from the following half cells, linked by a KCl salt bridge. • a Fe electrode in 1.0 M FeCl2 solution • a Ni electrode in 1.0
![PDF] Low-Temperature Specific Heat of Ni(NO3)2· 6H2O: Antiferromagnetic Interactions by A. Herweijer, S. A. Friedberg · 10.1103/physrevb.4.4009 · OA.mg PDF] Low-Temperature Specific Heat of Ni(NO3)2· 6H2O: Antiferromagnetic Interactions by A. Herweijer, S. A. Friedberg · 10.1103/physrevb.4.4009 · OA.mg](https://og.oa.mg/Low-Temperature%20Specific%20Heat%20of%20Ni(NO3)2%C2%B7%206H2O%3A%20Antiferromagnetic%20Interactions.png?author=%20A.%20Herweijer,%20S.%20A.%20Friedberg)
PDF] Low-Temperature Specific Heat of Ni(NO3)2· 6H2O: Antiferromagnetic Interactions by A. Herweijer, S. A. Friedberg · 10.1103/physrevb.4.4009 · OA.mg

An electrochemical cell is constructed using 1.0 M solutions of Fe(NO3)2 and Sn(NO3)2 and strips of Fe and Sn. A voltmeter is connected to the electrodes and a salt bridge connects the

SOLVED: These compounds are in water: State the compound that forms a precipitate or indicate 'no precipitate' NaCl + Ba(OH)2 AgCIO3 Mgl2 CaCl2 K2CO3 FeCl2 (NH4)2S04 Ni(NO3)2 KOH Ni(NO3)2 K25 Pb(NO3)2 K2CO3

Facile Hydrothermal Fabrication of an α-Ni(OH)2/N-Doped Reduced Graphene Oxide Nanohybrid as a High-Performance Anode Material for Lithium-Ion Batteries | Energy & Fuels
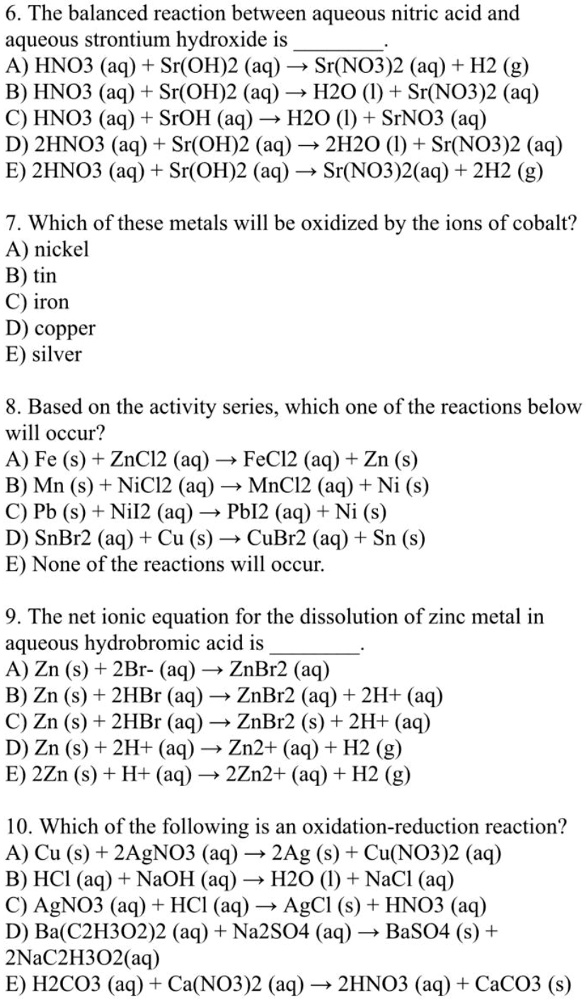
SOLVED: The balanced reaction between aqueous nitric acid and aqueous strontium hydroxide is HNO3 (aq) + Sr(OH)2 (aq) 57 Sr(NO3)2 (aq) + H2 (g) B) HNO3 (aq) + Sr(OH)2 (aq) 5 H2O (






![Synthesis of novel thiazolo[3,2‐a]chromeno[4,3‐d]pyrimidine‐6(7H)‐on Synthesis of novel thiazolo[3,2‐a]chromeno[4,3‐d]pyrimidine‐6(7H)‐on](https://www.researcher-app.com/image/eyJ1cmkiOiJodHRwczovL3MzLWV1LXdlc3QtMS5hbWF6b25hd3MuY29tL3N0YWNrYWRlbWljL3Byb2R1Y3Rpb24vcGFwZXIvNDAzMTI2Mi5wbmciLCJmb3JtYXQiOiJ3ZWJwIiwicXVhbGl0eSI6MTAwLCJub0NhY2hlIjp0cnVlfQ==.webp)