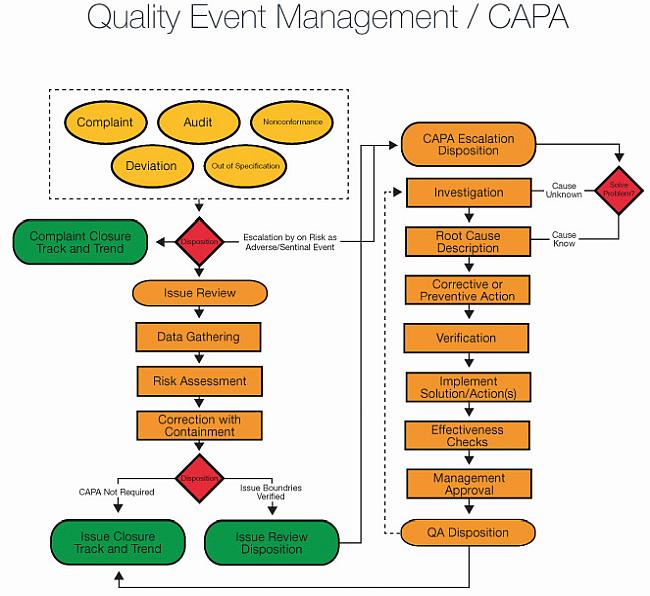
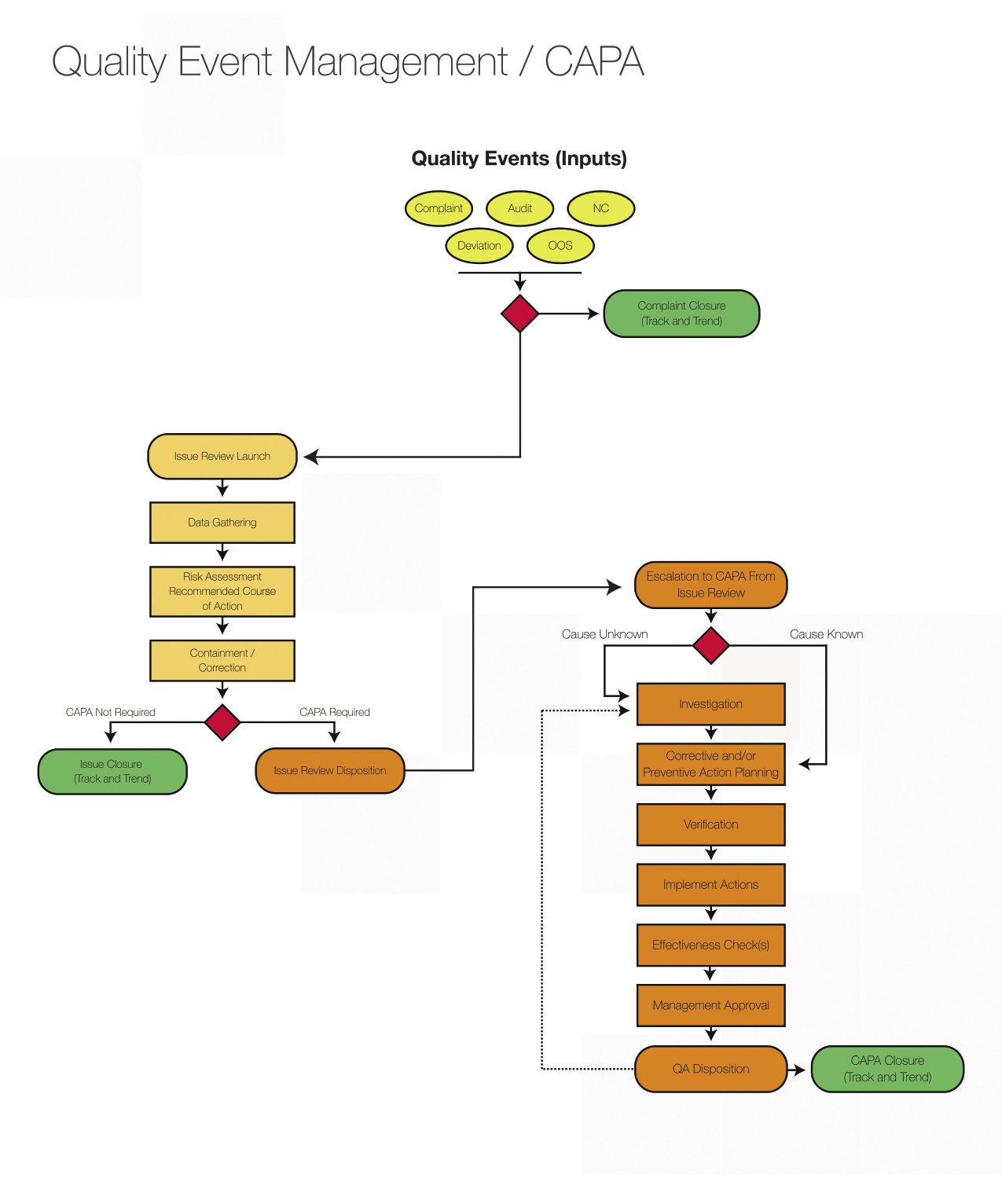
Amazon.com: Principles of corrective Action and Preventive Action :CAPA: A Handbook for Quality Professionals in Medical device and Pharmaceutical Industries eBook : MUCHEMU, DAVID N.: Kindle Store

Medical Device CAPA Process : Importance, benefits and implementation | Engineering - Johari Digital Healthcare Ltd.